Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (10): 1585-1590.doi: 10.3969/j.issn.2095-4344.2014.10.017
Previous Articles Next Articles
Creation and effectiveness of mammary stem cell medium
Zhang Jun-hong 1, 2, Yang Jing3, Dou Xiao-wei1, Wang Chun-hua4, Li Qing5, Zhao Chun-hua1
- 1 Center of Tissue Engineering, Department of Cell Biology, Basic Medical College of Peking Union Medical College & Basic Institute of Chinese Academy of Medical Science, Beijing 100005, China; 2 Department of Pathology, 4 Department of Otorhinolaryngology, Xingtai Eye Hospital, Xingtai 054000, Hebei Province, China; 3 Department of Physiology, Hebei Medical University, Shijiazhuang 050017, Hebei Province, China; 5 Department of Orthopaedics, Affiliated Hospital of Guiyang Medical College, Guiyang 550004, Guizhou Province, China
-
Online:2014-03-05Published:2014-03-05 -
Contact:Zhao Chun-hua, M.D., Professor, Doctoral supervisor, Center of Tissue Engineering, Department of Cell Biology, Basic Medical College of Peking Union Medical College & Basic Institute of Chinese Academy of Medical Science, Beijing 100005, China -
About author:Zhang Jun-hong, Attending physician, Center of Tissue Engineering, Department of Cell Biology, Basic Medical College of Peking Union Medical College & Basic Institute of Chinese Academy of Medical Science, Beijing 100005, China; Department of Pathology, Xingtai Eye Hospital, Xingtai 054000, Hebei Province, China -
Supported by:the Major State Basic Research Development Program of China (973 Program), No. 2011CB964900; the National High Technology Research and Development Program of China (863 Program), No. 2011AA020100; the Youth Foundation of Hebei Provincial Education Bureau, No. 2011154; the Scientific Supporting Plan of Xingtai City, No. 2013ZC062
CLC Number:
Cite this article
Zhang Jun-hong, Yang Jing, Dou Xiao-wei, Wang Chun-hua, Li Qing, Zhao Chun-hua. Creation and effectiveness of mammary stem cell medium[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(10): 1585-1590.
share this article
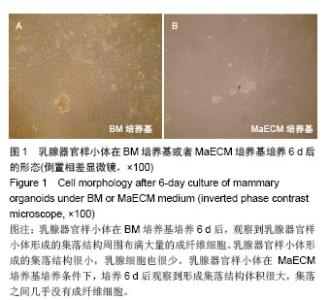
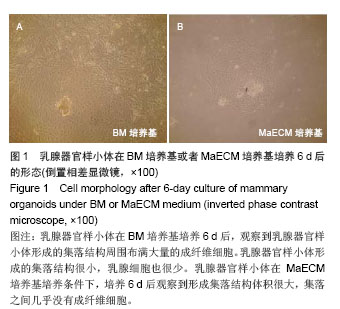
2.1 不同培养基对乳腺器官样小体培养后形态的影响 乳腺器官样小体在BM培养基培养6 d后,观察到乳腺器官样小体形成的集落结构周围布满大量的成纤维细胞。乳腺器官样小体形成的集落结构很小,乳腺细胞也很少(图1A)。在BM培养基中加入雌激素后,发现随着雌激素质量浓度的增加,成纤维细胞逐渐减少,集落结构逐渐变大,在1.5 μg/L的雌激素应用后基本没有观察到成纤维细胞,但是3 μg/L的雌激素导致集落结构丧失,细胞变性。在BM培养基中加入生长激素后,发现随着生长激素质量浓度的增加,没有改变成纤维细胞的生长,细胞生长速度增加。因此在含有BM培养基和1.5 μg/L雌激素培养基中加入生长激素,发现随着生长激素质量浓度的增加,可以明显促进集落的生长,但在应用100 μg/L的生长激素后出现类似3 μg/L雌激素的结果,包括集落结构丧失,细胞变性。因此,选择在BM培养基加入10 μg/L雌激素和50 μg/L生长激素做后续的实验,命名为MaECM (mammary epithelial cell medium)培养基。乳腺器官样小体在MaECM培养基培养条件下,培养6 d后观察到形成集落结构体积很大,集落之间几乎没有成纤维细胞(图1B)。"
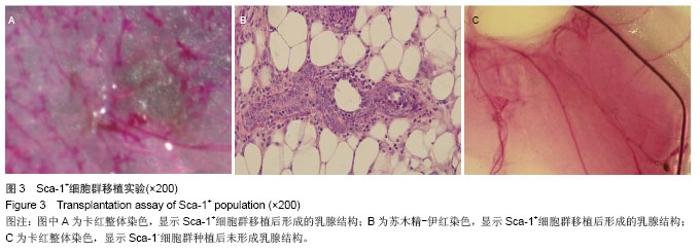
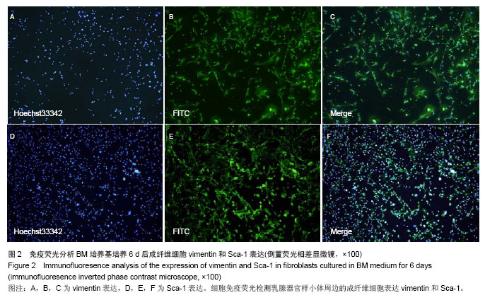
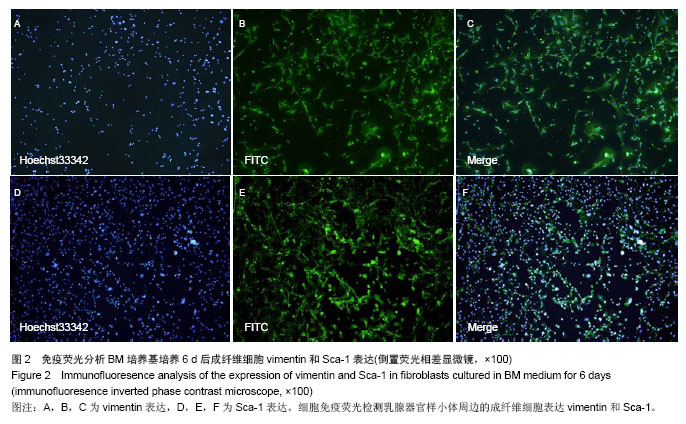
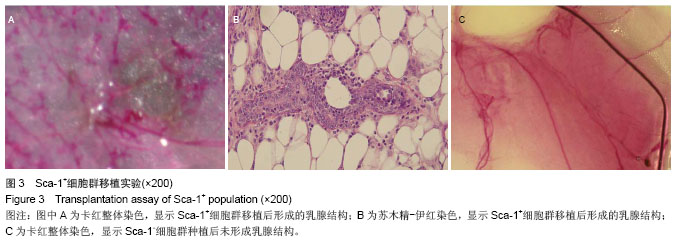
2.3 磁珠筛选Sca-1+和Sca-1-细胞群的纯度 乳腺器官样小体在MaECM培养基培养6 d后,经磁珠筛选和流式细胞术分析显示,Sca-1+细胞在Sca-1-和Sca-1+细胞群的比例分别是5%和92%。以前报道的Sca-1富集乳腺前体细胞的实验中,Sca-1+细胞在Sca-1+和Sca-1-乳腺群细胞的比例分别是86%和7%。因此磁珠筛选Sca-1+细胞在Sca-1+和Sca-1-乳腺细胞群的纯度可以满足后述的实验[24]。 2.4 Sca-1+细胞发育成乳腺结构的潜能 1×104 Sca-1+细胞种植在4只鼠双侧乳腺脂肪垫中,经过8-10周后,在种植的8个脂肪垫中,其中6个生长出乳腺结构。这些种植生成的乳腺结构和正常的乳腺结构相比,管泡状结构伸向不同的方向(图3A,B)。而在1×104 Sca-1-细胞种植组,其中一只鼠死亡,而在其余的6个脂肪垫中没有生长出乳腺结构(图3C)。脂肪垫清除实验是1959年以来证明乳腺干细胞最可靠的实验,以往文献说明在证实乳腺干细胞存在的实验中种植的乳腺细胞应少于2×104细胞[27-28]。因此,认为Sca-1+细胞具有富集乳腺干细胞的能力。"
| [1]Guo P, Huang Z, Yu P, et al. Trends in cancer mortality in China: an update. Ann Oncol. 2012; 23(10):2755-2762. [2]Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA: a cancer journal for clinicians. 2013; 63(1):11-30. [3]Nguyen LV, Vanner R, Dirks P, et al. Cancer stem cells: an evolving concept. Nat Rev Cancer. 2012; 12(2):133-143. [4]Tomasetti C, Vogelstein B, Parmigiani G. Half or more of the somatic mutations in cancers of self-renewing tissues originate prior to tumor initiation. Proc Natl Acad Sci U S A. 2013; 110(6):1999-2004. [5]Brenton JD, Stingl J, Stem cells: Anatomy of an ovarian cancer. Nature. 2013; 495(7440):183-184. [6]Hammoud SS, Cairns BR, Jones DA. Epigenetic regulation of colon cancer and intestinal stem cells. Curr Opin Cell Biol. 2013;25(2):177-183. [7]Holland JD, Klaus A, Garratt AN, et al. Wnt signaling in stem and cancer stem cells. Curr Opin Cell Biol. 2013;25(2):254- 264. [8]Günes C, Rudolph KL. The Role of Telomeres in Stem Cells and Cancer. Cell. 2013; 152(3):390-393. [9]Charafe-Jauffret E, Ginestier C, Bertucci F, et al. ALDH1-positive cancer stem cells predict engraftment of primary breast tumors and are governed by a common stem cell program. Cancer Res. 2013;73(24):7290-7300. [10]Kakarala M, Brenner DE, Korkaya H, et al. Targeting breast stem cells with the cancer preventive compounds curcumin and piperine. Breast Cancer Res Treat. 2010; 122(3): 777-785. [11]Han DW, Tapia N, Hermann A, et al. Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell. 2012; 10(4):465-472. [12]Chen J, McKay RM, Parada LF. Malignant glioma: lessons from genomics, mouse models, and stem cells. Cell. 2012; 149(1):36-47. [13]Amsterdam A, Raanan C, Schreiber L, et al. Differential localization of LGR5 and Nanog in clusters of colon cancer stem cells. Acta Histochemica. 2012. [14]Vizio B, Mauri FA, Prati A, et al. Comparative evaluation of cancer stem cell markers in normal pancreas and pancreatic ductal adenocarcinoma. Oncol Rep. 2012; 27(1):69-76. [15]Liang D, Shi Y. Aldehyde dehydrogenase-1 is a specific marker for stem cells in human lung adenocarcinoma. Med Oncol. 2012; 29(2):633-639. [16]Huch M, Dorrell C, Boj SF, et al. In vitro expansion of single Lgr5+ liver stem cells induced by Wnt-driven regeneration. Nature. 2013; 494(7436):247-250. [17]Rountree CB, Mishra L, Willenbring H. Stem cells in liver diseases and cancer: recent advances on the path to new therapies. Hepatology. 2012; 55(1):298-306. [18]Schwartz T, Stark A, Pang J, et al. Expression of aldehyde dehydrogenase 1 as a marker of mammary stem cells in benign and malignant breast lesions of Ghanaian women. Cancer. 2013; 119(3):488-494. [19]Smalley MJ, Kendrick H, Sheridan JM, et al. Isolation of mouse mammary epithelial subpopulations: a comparison of leading methods. J Mammary Gland Biol Neoplasia. 2012; 17(2):91-97. [20]Guler G, Balci S, Costinean S, et al. Stem cell-related markers in primary breast cancers and associated metastatic lesions. Mod Pathol. 2012; 25(7):949-955. [21]Sleeman KE, Kendrick H, Ashworth A, et al. CD24 staining of mouse mammary gland cells defines luminal epithelial, myoepithelial/basal and non-epithelial cells. Breast Cancer Res. 2005; 8(1):R7. [22]Shackleton M, Vaillant F, Simpson KJ, et al. Generation of a functional mammary gland from a single stem cell. Nature. 2006; 439(7072):84-88. [23]Liao MJ, Zhang CC, Zhou B, et al. Enrichment of a population of mammary gland cells that form mammospheres and have in vivo repopulating activity. Cancer Res. 2007; 67(17):8131- 8138. [24]Welm BE, Tepera SB, Venezia T, et al. Sca-1(pos) cells in the mouse mammary gland represent an enriched progenitor cell population. Dev Biol. 2002; 245(1):42-56. [25]Stingl J, Eirew P, Ricketson I, et al. Purification and unique properties of mammary epithelial stem cells. Nature. 2006; 439(7079):993-997. [26]McQualter JL, Brouard N, Williams B, et al. Endogenous Fibroblastic Progenitor Cells in the Adult Mouse Lung Are Highly Enriched in the Sca‐1 Positive Cell Fraction. Stem Cells. 2009; 27(3):623-633. [27]DeOme K, Faulkin L, Bern HA, et al. Development of mammary tumors from hyperplastic alveolar nodules transplanted into gland-free mammary fat pads of female C3H mice. Cancer Res. 1959; 19(5):515. [28]Regan J, Smalley M. Prospective isolation and functional analysis of stem and differentiated cells from the mouse mammary gland. Stem Cell Rev. 2007; 3(2):124-136. [29]Staszkiewicz J, Gimble JM, Manuel JA, et al. IFATS Collection: Stem Cell Antigen-1-Positive Ear Mesenchymal Stem Cells Display Enhanced Adipogenic Potential. Stem Cells. 2008; 26(10):2666-2673. [30]Kang SG, Shinojima N, Hossain A, et al. Isolation and perivascular localization of mesenchymal stem cells from mouse brain. Neurosurgery. 2010; 67(3):711. [31]Naftali‐Shani N, Itzhaki‐Alfia A, Landa‐Rouben N, et al. The Origin of Human Mesenchymal Stromal Cells Dictates Their Reparative Properties. J Am Heart Assoc. 2013; 2(5): e000253. [32]Tan KY, Eminli S, Hettmer S, et al. Efficient generation of iPS cells from skeletal muscle stem cells. PloS one. 2011; 6(10): e26406. [33]Suzuki K, Sun R, Origuchi M, et al. Mesenchymal stromal cells promote tumor growth through the enhancement of neovascularization. Molecular Med. 2011; 17(7-8):579. [34]Siegel G, Krause P, Wöhrle S, et al. Bone marrow-derived human mesenchymal stem cells express cardiomyogenic proteins but do not exhibit functional cardiomyogenic differentiation potential. Stem Cells Dev. 2012; 21(13): 2457-2470. [35]Sakakura T, Suzuki Y, Shiurba R. Mammary stroma in development and carcinogenesis. J Mammary Gland Biol Neoplasia. 2013:1-9. [36]dos Santos CO, Rebbeck C, Rozhkova E, et al. Molecular hierarchy of mammary differentiation yields refined markers of mammary stem cells. Proc Natl Acad Sci. 2013; 110(18): 7123-7130. [37]Roy S, Gascard P, Dumont N, et al. Rare somatic cells from human breast tissue exhibit extensive lineage plasticity. Proc Natl Acad Sci. 2013; 110(12):4598-4603. [38]Fan Y, Menon RK, Cohen P, et al. Liver-specific deletion of the growth hormone receptor reveals essential role of growth hormone signaling in hepatic lipid metabolism. J Biol Chem. 2009; 284(30):19937-19944. [39]Horigan K, Trott J, Barndollar A, et al. Hormone interactions confer specific proliferative and histomorphogenic responses in the porcine mammary gland. Domest Anim Endocrinol. 2009; 37(2):124-138. [40]Cheng G, Weihua Z, Warner M, et al. Estrogen receptors ER alpha and ER beta in proliferation in the rodent mammary gland. Proc Natl Acad Sci U S A. 2004; 101(11):3739-3746. [41]Morani A, Warner M, Gustafsson JA. Biological functions and clinical implications of oestrogen receptors alfa and beta in epithelial tissues. J Intern Med. 2008; 264(2):128-142. [42]Clarke RB, Spence K, Anderson E, et al. A putative human breast stem cell population is enriched for steroid receptor-positive cells. Dev Biol. 2005; 277(2):443-456. [43]Booth BW, Smith GH. Estrogen receptor-alpha and progesterone receptor are expressed in label-retaining mammary epithelial cells that divide asymmetrically and retain their template DNA strands. Breast Cancer Res. 2006; 8(4): R49. [44]Booth BW, Boulanger CA, Smith GH. Selective segregation of DNA strands persists in long-label-retaining mammary cells during pregnancy. Breast Cancer Res. 2008; 10(5):R90. [45]Wang S, Qu X, Zhao RC. Clinical applications of mesenchymal stem cells. J Hematol Oncol. 2012; 5(1):19. |
| [1] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [2] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [3] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [4] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [5] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [6] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [7] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [8] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [9] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [10] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [11] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [12] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [13] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [14] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [15] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||